What are cephalosporins?
Cephalosporins are a class of β-lactam antibiotics. It is originally derived from the fungus Cephalosporium acremonium and previously called Cephalosporium.
Cephamycins is a methoxy group at position 7 of the beta-lactam ring (7-aminocephalosporanic acid nucleus) and together with cephamycins constitute a subgroup of β-lactam antibiotics called cephems.
History and Source
The first source of the cephalosporins isolated from Cephalosporium acremonium. It was isolated in 1948 by Italian scientist Giuseppe Brotzu from the sea near a sewer outlet off the Sardinian coast.
The first agent cephalothin (cefalotin) was launched by the scientist Eli Lilly in 1964, With the isolation of the active nucleus of ‘cephalosporin-C ‘containing 7-aminocephalosporanic acid and with the addition of side chains of semisynthetic compounds.
Chemistry of cephalosporins
The compounds contain 7-aminocephalosporanic acid which is relatively stable in dilute acid and highly resistant to penicillinase.
The modifications at position 7 of the β-lactam ring are associated with alteration in antibacterial activity. The substitutions at position 3rd of the dihydrothiazine ring and it is associated with changes in the metabolism & pharmacokinetic properties of the drugs.
Classification of cephalosporins
Based on their chemical structure, clinical pharmacology, resistance to b-lactamase or antimicrobial spectrum, and nowadays a well-accepted system of classification by “generations” which is very useful. These drugs only used after doctor or consultant prescriptions.
- First-generation cephalosporins
- Cefazolin (IM, IV)
- Cephalexin(O)
- Cephadroxil(O)
- Cephalothin (IM)
- Cephradine(O, IM, IV)
- Second-generation cephalosporins
- Cefuroxime (O)
- Cefaclor (O)
- Cefprozil (O)
- Cephamycins:
- Cefoxitin (IM, IV)
- Cefotetan (IM)
- Cefmetazole
- Third generation cephalosporins
- Cefotaxime (IM, IV)
- Ceftriaxone(IM, IV)
- Ceftazidime(IM, IV)
- Cefoperazone(IM, IV)
- Cefixime (O)
- Cefpodoxime(O)
- Fourth-generation cephalosporins
- Cefepime (IV)
- Cefpirome(IM, IV)
- Fifth-generation cephalosporins
- Ceftobiprole
- Ceftaroline
First- generation
They have good activity against gram-positive bacteria & relatively modest activity against gram-negative microorganisms.
Most of the gram-positive cocci with some exceptions such as enterococci, methicillin-resistant S. aureus, and S. epidermidis are susceptible.
Most oral cavity anaerobic organisms are sensitive, but the B. fragilis group is resistant. The activity against Moraxella catarrhalis, E. coli, K. pneumoniae, and P. mirabilis (PEK-M) is good.
Second generation
The 2nd generation of cephalosporins increased their activity against gram-negative microorganisms such as HENPEK organisms (H. influenzae, Enterobacter, N. gonorrhoeae, Proteus, E. coli, K. pneumoniae).
It is less active than the third-generation agents. A subset of second-generation agents (cefoxitin, cefotetan, and cefmetazole) also active against the B. fragilis group of organisms.
Third generation
Generally are less active than first-generation agents against gram-positive cocci nut more active against the Enterobacteriaceae, including beta-lactamase-producing strains of the organisms.
A subset of third-generation agents (ceftazidime and cefoperazone) also active against Pseudomonas aeruginosa and less active than other third-generation agents against gram-positive cocci.
Fourth generation
An extended spectrum of activity against Gram-positive (+) bacilli compared with the third-generation agents. The fourth-generation agents increased stability from hydrolysis by plasmid and chromosomally mediated b-lactamases.
Fourth-generation agents are particularly useful in the empirical treatment of serious infections in hospitalized patients, gram-positive microorganisms, Enterobacteriaceae, and Pseudomonas.
Fifth-generation
It is a broad-spectrum, advanced generation drug that is active against MRSA and streptococcus pneumoniae & enterococcus. Gram-negative activity is similar to the 3rd generation.
P. aeruginosa, extended-spectrum β- lactamase (ESBL)-producing Enterobacteriaceae, and Acinetobacter baumannii. The 5th generation cephalosporins are of limited activity against anaerobes.
Mechanism of cephalosporins
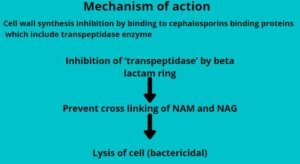
The cell wall synthesis inhibition by binding to cephalosporins binding proteins (CBPs) which include transpeptidase enzymes.
The inhibition of ‘transpeptidase’ by beta-lactam ring prevents cross-linking of NAM and NAG and finally Lysis of the cell (bactericidal).
Mechanisms of Bacterial Resistance
- Inability to reach the site of action due to Impermeability to the antibiotic or efflux so that it does not reach its site of action.
- Alteration in target proteins and decrease the affinity of the drug.
- Elaboration of beta-lactamases (cephalosporinase) that destroy specific cephalosporins.
Pharmacokinetics of cephalosporins
- Route of Administration: oral, IM,IV
- Distribution:
- CSF, placenta, synovial and pericardial fluids, aqueous humor of eye, bile
- Excretion:
- Kidney(dosage thus should be altered in patients with renal insufficiency)
Uses of Cephalosporins
- Upper respiratory tract infections
- Cutaneous and soft tissue infections
- ENT infections
- Urinary tract infections
- Septicaemias caused by Gram-negative organism
- Surgical prophylaxis
- Meningitis
- Gonorrhea
- Chancroid
- Typhoid
- Mixed aerobic anaerobic infections
- Hospital-acquired infections
- Community-acquired pneumonia
- Prophylaxis & treatment of infections in neutropenic patients
Adverse effects of Cephalosporins
- Hypersensitivity reactions:
- Anaphylaxis, bronchospasm, urticaria, rashes
- Cross-reactivity to penicillin is seen in some patients.
- Pain after injection, thrombophlebitis
- Diarrhea, vomiting, and anorexia
- Nephrotoxicity( cephaloridine)
- Bleeding (cefoperazone) can occur either due to hypoprothrombinemia or thrombocytopenia and or platelet dysfunction.
- Neutropenia, thrombocytopenia
- Disulfiram like interaction(cefoperazone)
- Positive coomb’s test

exelent notes